Atomic emission spectrum quizlet12/3/2023  Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. The emission spectrum of a chemical element is the pattern of wavelengths obtained when the element is subjected to a specific excitation. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. (b) Images of the emission and absorption spectra of hydrogen are shown here. 
(a) When a hydrogen atom absorbs a photon of light, an electron is excited to an orbit that has a higher energy and larger value of n. The Figure below shows the atomic emission spectrum of hydrogen. 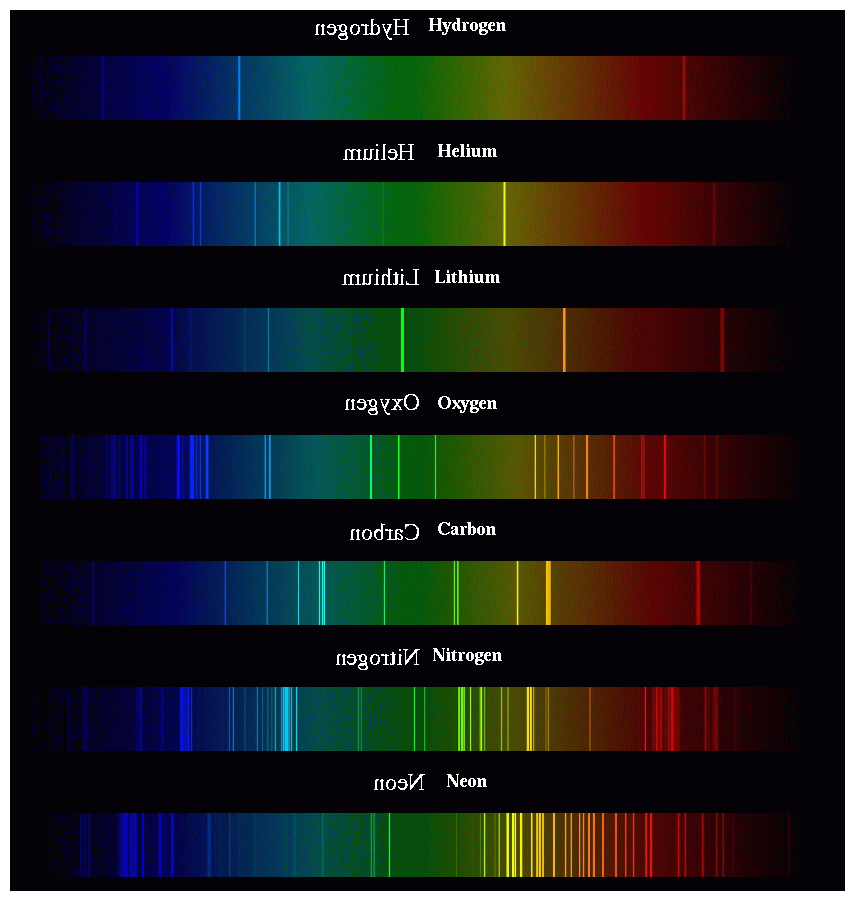 
In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. The atomic emission spectrum is the pattern formed when light passes through a prism or grating to separate it into the different of light it. Figure 7.3.6: Absorption and Emission Spectra. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. a single wavelength of light that can be seen when the light from an excited element is passed through a prism Spectrum. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
